
Using a battery desulphator
by
A battery desulphator is a device that is primarily meant for preventing the
build-up of sulphur precipitate inside of a lead acid battery. Sulphation is
common in lead acid batteries, particularly if the battery has been left unattended
for an extended period of time. There is also a possible benefit from using
a desulphator on healthy batteries or batteries that have reduced capacity from
age. I recently tried using desulphator on my aging pack of US-2200 batteries.
The desulphator I used is a Solar Converters, Inc model BD-2 that I borrowed
from John Schaefer. This particular model can be adjusted to work from 10 to
60 volts and according to the label draws about 50mA. Since my batteries are
6 volts, I had to use two batteries in series to operate the desulphator. This
was not a problem since I could easily find two adjacent batteries in my pack
that had suspicious readings on the volt meter and hydrometer. I left the desulphator
on for over 24 hours, while a small power supply provided a trickle charge of
about 2 amps into the batteries.
After running the desulphator and the charger for several days on four different
batteries, I checked the readings again. One of the batteries that had previously
had poor readings seemed to be much better, while another battery did not seem
to improve at all. It is hard to say whether the desulphator made much of a
difference, but it certainly didn't do any harm!
In order to see what the desulphator was doing, I hooked up my oscilloscope
while it was operating. When I took a direct reading of the voltage, nothing
was visible on the scope, but by hooking up a small length of wire as a shunt,
I was able to see the current. The wave form appeared to be a sharp spike followed
by a small "bounce."
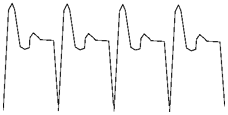
The frequency of the waveform was approximately 1kHz. I would speculate that
the desulphator is best at it's intended purpose of preventing sulphur build-up
in battery cells. In the case of my aging batteries, I think the desulphator
may be somewhat beneficial, but the batteries are nearing the end of their life
cycle, and it is unlikely that anything can delay their impending failure.
